 Because of this, they are rarely found in elemental form, and are instead most often found in compounds with other elements. 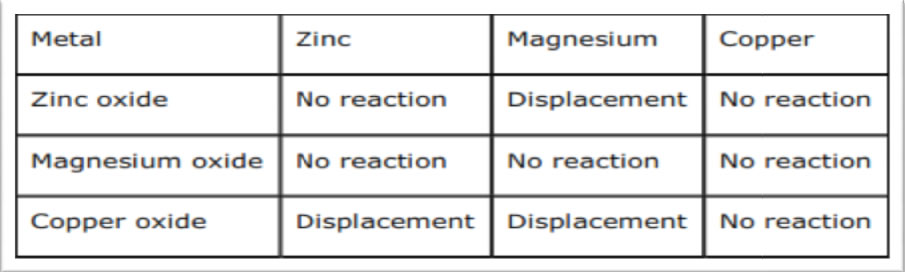
They are highly electropositive - meaning they have a tendency to give away their valence electron. The Alkali metals are the most reactive metals in the periodic table. Alkali metals, particularly sodium, are important in commercial use and chemical synthesis.Īlkali metals are highly conductive soft metals, which have a high lustre that oxidizes quickly when they are exposed to air. For example, potassium hydroxide, or lithium hydroxide.Īlkali metals have been studied since 1807, when Sir Humphry Davy explored the electrical properties of potassium and sodium. They are called alkali metals because they react strongly with water to form alkalies - hydroxide compounds made up of the element plus an -OH (hydroxide). Why are the group one elements called alkali metals? The alkali metals are located in the first column of the periodic table and include all the elements in that column except for hydrogen.ģ. Where are the alkali metals located on the periodic table? Thus, this electron can easily escape the positive pull of the nucleus to be donated to other elements, resulting in a reaction.Ģ. FAQsĪlkali metals are so reactive due to their electronic configuration - they have one valence electron and a large atomic radius. Metals make up almost the entire left side of the periodic table, save for Hydrogen which is a nonmetal. Elements in the periodic table can be described as metals, metalloids, and nonmetals. Elements in the same group typically have similar chemical properties as a result of their similar electronic configuration. All of the elements in a group share the same number of valence electrons: electrons in their outermost shell. 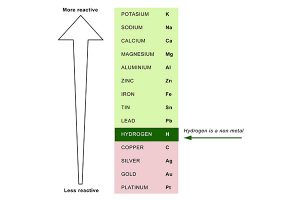
Refresher: The periodic table is organized into groups - where each column comprises a group. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
